2024 | Clonal haematopoiesis is associated with major adverse cardiovascular events in patients with hypertrophic cardiomyopathy
https://pmccbiobank.ca/wp-content/themes/osmosis/images/empty/thumbnail.jpg 150 150 fraser.amos@uhn.ca fraser.amos@uhn.ca https://secure.gravatar.com/avatar/4700def0819703d34d07e41db7e99c3af5d01396f322703ebefce3c89a1de6e3?s=96&d=mm&r=g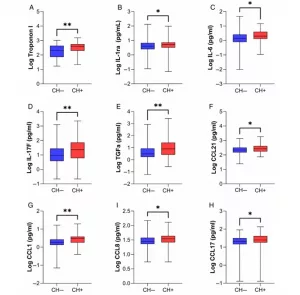 Authors: Fernando L. Scolari, Darshan Brahmbhatt, Sagi Abelson, Deacon Lee, Raymond H. Kim, Ali Pedarzadeh, Ali Sakhnini, Arnon Adler, Raymond H. Chan, John E. Dick, Harry Rakowski, and Filio Billia
Authors: Fernando L. Scolari, Darshan Brahmbhatt, Sagi Abelson, Deacon Lee, Raymond H. Kim, Ali Pedarzadeh, Ali Sakhnini, Arnon Adler, Raymond H. Chan, John E. Dick, Harry Rakowski, and Filio Billia
Short Description: Hypertrophic cardiomyopathy (HCM) is an inherited cardiovascular disorder, affecting 1 in 500 individuals. However, germline pathogenic or likely pathogenic (P/LP) genetic variants, such as MYH7 and MYBPC3 genes, are identified in only 25–40% of patients. A wide heterogeneity and distinct morphologies of left ventricular (LV) hypertrophy are described, even amongst family members carrying similar genetic variants. Interestingly, interstitial fibrosis is detected in 40–70% of patients and is associated with sudden cardiac death (SCD). Although HCM can be well tolerated, a subset of patients may require advanced treatments such as orthotopic heart transplant (OHT) and LV assist devices. The underlying mechanisms involved in the heterogeneous clinical presentation of HCM patients are not well understood. Clonal haematopoiesis (CH) has emerged as a new risk factor for the development of atherosclerosis, heart failure and cardiogenic shock and is associated with worse outcomes.
Interest: Cardiac magnetic resonance imaging, Clonal haematopoiesis, Fibrosis, Hypertrophic cardiomyopathy, Inflammation