2023| Myocardial Inflammation at FDG PET/MRI and Clinical Outcomes in Symptomatic and Asymptomatic Participants after COVID-19 Vaccination
https://pmccbiobank.ca/wp-content/themes/osmosis/images/empty/thumbnail.jpg 150 150 fraser.amos@uhn.ca fraser.amos@uhn.ca https://secure.gravatar.com/avatar/4700def0819703d34d07e41db7e99c3af5d01396f322703ebefce3c89a1de6e3?s=96&d=mm&r=g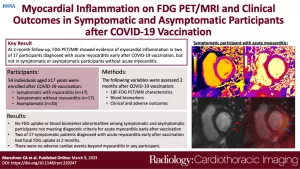
Authors: Constantin Arndt Marschner, Paaladinesh Thavendiranathan, Dakota Gustafson, Kathryn L. Howe, Jason E. Fish, Robert M. Iwanochko, Rachel M. Wald, Husam Abdel-Qadir, Slava Epelman, Angela M. Cheung, Rachel Hong, Kate Hanneman
Short Description: As of August 2022, more than 5.3 billion people worldwide have received at least one dose of a COVID-19 vaccine (1). Several adverse events have been reported, including myocarditis and pericarditis, following the administration of mRNA-based COVID-19 vaccines (2,3). The overall incidence of myopericarditis following COVID-19 vaccines is low, estimated at 18 per 1 million vaccine doses, with the highest risk in adolescent and young adult males (4). However, many more patients experience cardiac symptoms after vaccination, including shortness of breath, palpitations, and chest pain, yet do not meet diagnostic criteria for acute myopericarditis (5). The cause of these symptoms and the natural history of these patients remains unknown.
Cardiac MRI plays an important role in the assessment of myocardial tissue changes (6). Cardiac fluorine 18 (18F) fluorodeoxyglucose (FDG) PET provides complementary physiologic information, allowing for the assessment of changes in myocardial metabolism (7,8). Several recent case series have reported cardiac MRI findings in patients with myocarditis following COVID-19 vaccination (9,10). However, there are limited data on patients who present with new-onset cardiac symptoms following COVID-19 vaccination but do not meet diagnostic criteria for myopericarditis (5). There are also limited data evaluating potential subclinical myocardial tissue changes in asymptomatic individuals after vaccination.
The purpose of this study was to identify cardiac sequelae of COVID-19 vaccination and relate patient-reported cardiac symptoms to myocardial tissue changes identified with cardiac FDG PET/MRI, circulating biomarkers of cardiac injury and systemic inflammation, health-related quality of life, and adverse cardiac events at 2-month follow-up. Our goal was to inform guidelines for cardiac investigations after COVID-19 vaccination and the potential need for longer-term follow-up.
Interest: Myocardial Inflammation, COVID-19, Vaccination, Molecular Imaging, MR Imaging